By Diepreye Robinson Erebi
Thank you for reading this post, don't forget to subscribe!Introduction
Polycystic Ovary Syndrome (PCOS) is one of the most prevalent and complex endocrine-metabolic disorders affecting women of reproductive age, with a global prevalence estimated at 6-15% (Bozdag et al., 2016), and potentially higher when undiagnosed cases are considered. This multifactorial condition is characterized by a combination of hyperandrogenism, ovulatory dysfunction, and polycystic ovarian morphology(PCOM).

Polycystic ovary syndrome(PCOS) has significant implications not only for reproductive health but also for metabolic and cardiovascular risk. While much of the focus in PCOS management is on hormonal and metabolic factors, anatomical changes or alterations, particularly in the ovarian structural changes, are critical to its diagnosis and pathophysiology. Understanding the anatomical alterations provides valuable insights into the mechanisms of the syndrome and informs both diagnostic and therapeutic approaches. These anatomical deviations are intricately linked to disrupted folliculogenesis, excessive androgen production, and persistent anovulation.
According to the Rotterdam criteria, a diagnosis of PCOS requires at least two of the following three features: oligo- or anovulation, clinical or biochemical signs of hyperandrogenism, and polycystic ovaries visible on ultrasound (Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group, 2004).
What is Polycystic Ovary Syndrome ?
Polycystic ovary syndrome (PCOS) is a hormonal imbalance that occurs when the ovaries (the organ that produces and releases eggs) create excess hormones. In thus condition, the ovaries produce unusually high levels of hormones called androgens- a masculinizing hormone. This causes the reproductive hormones to become imbalanced. As a result, people with PCOS often have irregular menstrual cycles, missed periods and unpredictable ovulation. Small follicle cysts (fluid-filled sacs with immature eggs) may be visible on the ovaries on ultrasound due to lack of ovulation (anovulation). However, despite the name “polycystic,” the condition can still be diagnosed in the absence of cysts. The ovarian cysts aren’t dangerous or painful.
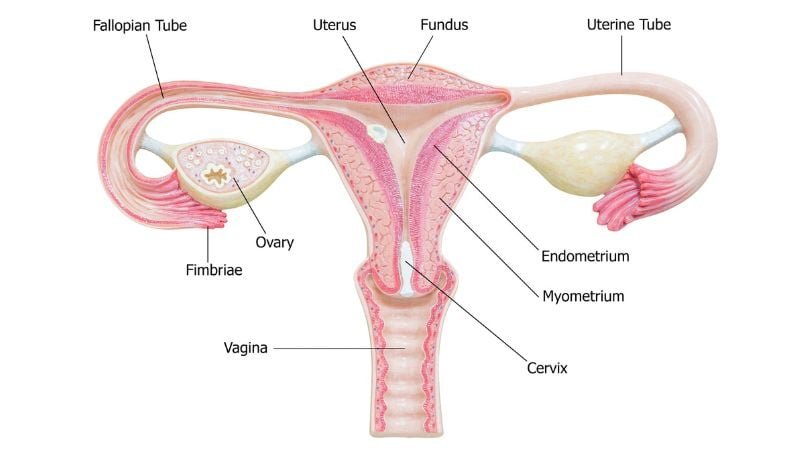
Normal Anatomy of the Female Reproductive System
The female reproductive system includes the ovaries, fallopian tubes, uterus, cervix, and vagina. The ovaries are almond-shaped structures located in the lateral wall of the pelvis. They are situated on either side of the uterus and are responsible for producing oocytes and hormones like estrogen and progesterone. It is connected to the uterus via the fallopian tube.
Each ovary has an outer cortex, which contains developing follicles at various stages, and a central medulla composed of vascularized connective tissue, blood vessels, and lymphatics. The development and release of oocytes are regulated by the hypothalamic-pituitary-ovarian (HPO) axis. Gonadotropin-releasing hormone (GnRH) from the hypothalamus stimulates the anterior pituitary to release luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which act on ovarian follicles.These anatomical structures play critical roles in menstruation, ovulation, and fertility (Marieb & Hoehn, 2019).
Follicular development occurs in stages: primordial, primary, secondary, tertiary (Graafian), and ovulation. Normally, one dominant follicle matures and ovulates in each cycle. Post-ovulation, the ruptured follicle becomes the corpus luteum, which secretes progesterone.
Anatomical Changes in PCOS
PCOS is associated with distinctive anatomical changes in the ovaries that are visible through imaging and observable in histological studies.
- Ovarian Enlargement: Ovaries in PCOS are typically 1.5 to 3 times larger than normal due to the accumulation of multiple immature follicles and stromal hypertrophy. An ovarian volume greater than 10ml per ovary is also indicative of PCOS (WHO., 1991).
- Thickened Tunica Albuginea: The fibrous outer capsule becomes denser and contributes to resistance to follicular rupture, which may contribute to anovulation.
- Increased Ovarian Stroma: A significant increase in the volume and echogenicity of the stromal tissue is commonly observed. The stroma is the site of androgen synthesis, and its hypertrophy is associated with hyperandrogenism.
- Multiple Small Follicles: More than 12 follicles measuring 2–9 mm in diameter may be observed peripherally in the ovary. This is referred to as the “string of pearls” sign on ultrasound imaging.
- Absence of Dominant Follicle/Follicular Arrest: Despite the presence of numerous small follicles, no follicle reaches the size required for ovulation due to elevated anti-Müllerian hormone (AMH) (Dewailly et al., 2016).
Pathophysiological Mechanisms Related to Anatomy
IThe anatomical changes in PCOS are closely associated with underlying hormonal imbalances and metabolic dysregulation.
- Disrupted Folliculogenesis: Due to elevated luteinizing hormone(LH) levels and altered luteininzing hormone to follicle stimulating hormone(FSH) ratio, follicular maturation is arrested. The follicles fail to reach the pre-ovulatory stage.
- Theca Cell Hyperplasia: LH overstimulation leads to proliferation of theca cells in the ovarian stroma, increasing androgen production (e.g., testosterone, androstenedione).
- Anovulation: The absence of a dominant follicle prevents ovulation, leading to irregular or absent menstruation.
- Insulin Resistance: Common in PCOS, insulin resistance exacerbates androgen production by acting synergistically with LH. It also impacts follicular development and promotes stromal hypertrophy.
- Hyperandrogenism: Theca cells overproduce androgens due to LH hypersecretion and insulin resistance (Azziz et al., 2016). High androgen levels prevent the ovaries from releasing eggs, which causes irregular menstrual cycles. Irregular ovulation can also cause small, fluid-filled sacs to develop on the ovaries. High androgen also causes acne and excess hair growth in females.
- Genetic and Epigenetic Influences: Family history and gene-environment interactions may influence ovarian morphology and hormone regulation.
Anatomical And Endocrine Changes
The anatomical changes in the ovaries are linked with dysregulation in the hypothalamic-pituitary-ovarian axis. Increased luteinizing hormone (LH) secretion, in relation to follicle-stimulating hormone (FSH), promotes androgen production by theca cells in the ovary.
These androgens can then be converted to estrogens in peripheral tissues, leading to disrupted folliculogenesis. The thickened ovarian capsule may also contribute to mechanical anovulation (Norman et al., 2007).
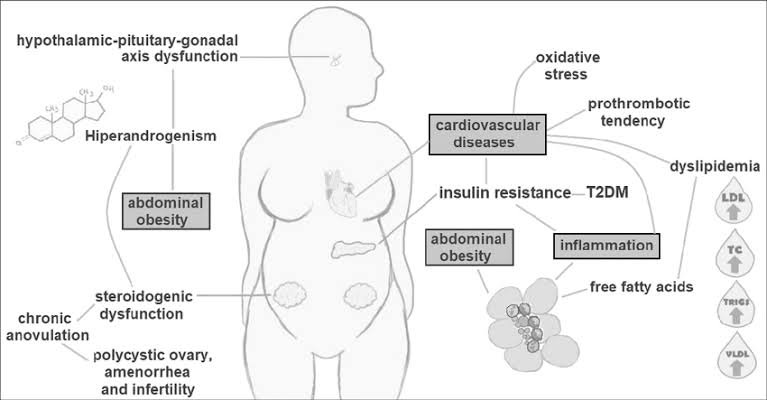
Important biochemical pathways involved in the pathophysiology of PCOS. Hyperandrogenism is a central feature in PCOS. Patients have a dysfunction in the hypothalamic-pituitary-gonadal axis, which influences steroidogenesis. In the ovaries, theca cells exhibit steroidogenic dysregulation that elevates circulating androgens. In addition, women with PCOS have lower levels of SHBG, which raise the level of free testosterone. Hormonal imbalance causes the follicular development to be prematurely disrupted, causing chronic anovulation, amenorrhea, polycystic ovaries and infertility.
Hyperandrogenism is also associated with accumulation of fat in the abdominal region and hyperinsulinemia secondary to insulin resistance (IR). Inflammation is considered an important link between the metabolic effects of PCOS, such as IR, dyslipidemia and T2DM. Visceral obesity causes an increase in plasma levels of inflammatory mediators and the adipocytes release fatty acids by lipolysis, causing dyslipidemia. PCOS women have higher oxidative stress markers and also an imbalance between pro-and anti-coagulant mediators.
Hemostatic and oxidative imbalances, combined with inflammation, IR and dyslipidemia arefactors that increase cardiovascular risk in these patients. TC = total cholesterol, Trigs = triglycerides, T2DM = type 2 diabetes mellitus.
Symptoms of Polycystic Ovary Syndrome
● Irregular periods or amenorrhea
● Abnormal hair growth (Hirsutism)
● Acne
● Skin tags
● Darkening of Skin
● Obesity
● Thinning of hair
● Ovarian cysts
● Infertility
It is also possible to suffer from Polycystic Ovary Syndrome and not have any symptoms as many women are only made aware of this condition when they experience difficulty getting pregnant and gain weight for unknown reasons.
Having PCOS may also increase the risk for certain pregnancy complications, although most people with PCOS are able to successfully carry a pregnancy. Other complications of PCOS
related to pregnancy include increased risk of:
● Gestational diabetes, preeclampsia and high blood pressure.
● Preterm birth (birth before 37 weeks of pregnancy) or C-section delivery due to obesity,
diabetes or high blood pressure.
Clinical Correlation
The anatomical changes of PCOS have a wide range of clinical implications, extending from reproductive to systemic manifestations.
- Menstrual Irregularities: Women with PCOS often experience oligomenorrhea (infrequent menstruation) or amenorrhea (absence of menstruation) due to anovulation.
- Hyperandrogenism: Elevated androgens result in clinical signs such as acne, hirsutism (excessive hair growth), and androgenic alopecia (hair thinning).
- Infertility: One of the most common causes of infertility, PCOS disrupts normal ovulation, making conception difficult without intervention.
- Obesity and Metabolic Syndrome: Many women with PCOS have central obesity, insulin resistance, dyslipidemia, and an increased risk of type 2 diabetes.
- Endometrial Hyperplasia and Cancer: Chronic anovulation leads to unopposed estrogen exposure, increasing the risk of endometrial hyperplasia and potentially endometrial carcinoma.
- Psychological Impact: Depression, anxiety, and low self-esteem are common due to the chronic nature of the disorder and its visible symptoms.
Imaging and Diagnostic Tools
- Transvaginal Ultrasonography (TVUS): It is the primary imaging tool used for the diagnosis of PCOS as it allows for high-resolution images of the ovaries. It is preferred due to its ability to clearly visualize ovarian morphology, including the number and size of follicles and the volume of the ovaries (Balen et al., 2016). A typical finding in PCOS is the presence of 12 or more follicles per ovary, measuring 2–9 mm in diameter, or an increased ovarian volume greater than 10ml (Teede et al., 2018).
In adolescent girls or in women where transvaginal ultrasound is not feasible, transabdominal ultrasonography may be used, though it is less sensitive.
• Diagnostic Criteria: The Rotterdam criteria require two of the following: oligo/anovulation, hyperandrogenism (clinical or biochemical), and polycystic ovaries on ultrasound.
*Oligo-ovulation or anovulation: Irregular or absent menstrual periods, indicating infrequent or absent ovulation.
*Clinical and/or biochemical signs of hyperandrogenism: This includes symptoms such as hirsutism (excess hair growth), acne, or male-pattern baldness, and/or elevated levels of androgens (male hormones) in blood tests.
*Polycystic ovaries on ultrasound: One or both ovaries having 20 or more follicles measuring 2–9 mm in diameter, or increased ovarian volume (>10 ml).
Recent Advances In Anatomical Research on PCOS Imaging Innovations
- 3D Ultrasound & Doppler: Detects increased stromal blood flow, linked to hyperandrogenism (Lam et al., 2019).
- MRI Elastography: Measures ovarian stiffness caused by fibrosis (Meyer et al., 2021).
Genetic And Molecular Insights
- Single-Cell RNA Seq: Reveals theca cell overactivity in androgen production (Liu et al., 2022).
- MicroRNAs: miR-21 (fibrosis) and miR-27a (aromatase suppression) disrupt folliculogenesis (Roth et al., 2020).
Stem Cell Research
- Ovarian Stem Cells: Potential to restore follicle development in PCOS (Wagner et al., 2020).
Gut-Ovary Axis
- Dysbiosis: Gut inflammation exacerbates insulin resistance and hyperandrogenism (Torres et al., 2020).
Treatment Implications Based on Anatomy
An understanding of the anatomical basis of PCOS allows for more targeted therapeutic strategies.
- Lifestyle Modifications: Weight reduction through diet and exercise can significantly reduce ovarian volume, improve insulin sensitivity, and restore ovulation.
- Hormonal Therapies: Oral contraceptives regulate menstrual cycles and reduce hyperandrogenic symptoms by suppressing LH and ovarian androgen production.
- Insulin-Sensitizing Agents: Metformin improves insulin resistance and may indirectly restore ovulation.
- Ovulation Induction: Clomiphene citrate, letrozole, or gonadotropins are used to stimulate follicular development in women seeking fertility.
- Surgical Intervention: Laparoscopic ovarian drilling (LOD) is a procedure that reduces androgen-producing stroma, leading to ovulation in some patients.
- In Vitro Fertilization (IVF): Used in cases where other methods fail, especially in the presence of anatomical barriers to conception.
*Seminar Presentation by Diepreye Robinson Erebi (UG/20/0990), Faculty of Basic medical Sciences, Department of Human Anatomy, Niger Delta University, Amassoma, Bayelsa State
Kindly share





